Transdermal Nanomedicine: Pharmaceutical Advances for Controlled and Painless Drug Delivery
DOI:
https://doi.org/10.64474/3107-6386.Vol2.Issue1.4Keywords:
- Transdermal Drug Delivery, Nanocarriers, Nanomedicine, Skin Penetration, Controlled Release, Liposomes, Nanostructured Lipid Carriers, Polymeric Nanoparticles, Bioavailability, Transdermal Patches, Microneedles, Pharmacokinetics, Targeted Delivery, Patient Compliance, Theranostic Nanomedicine
Abstract
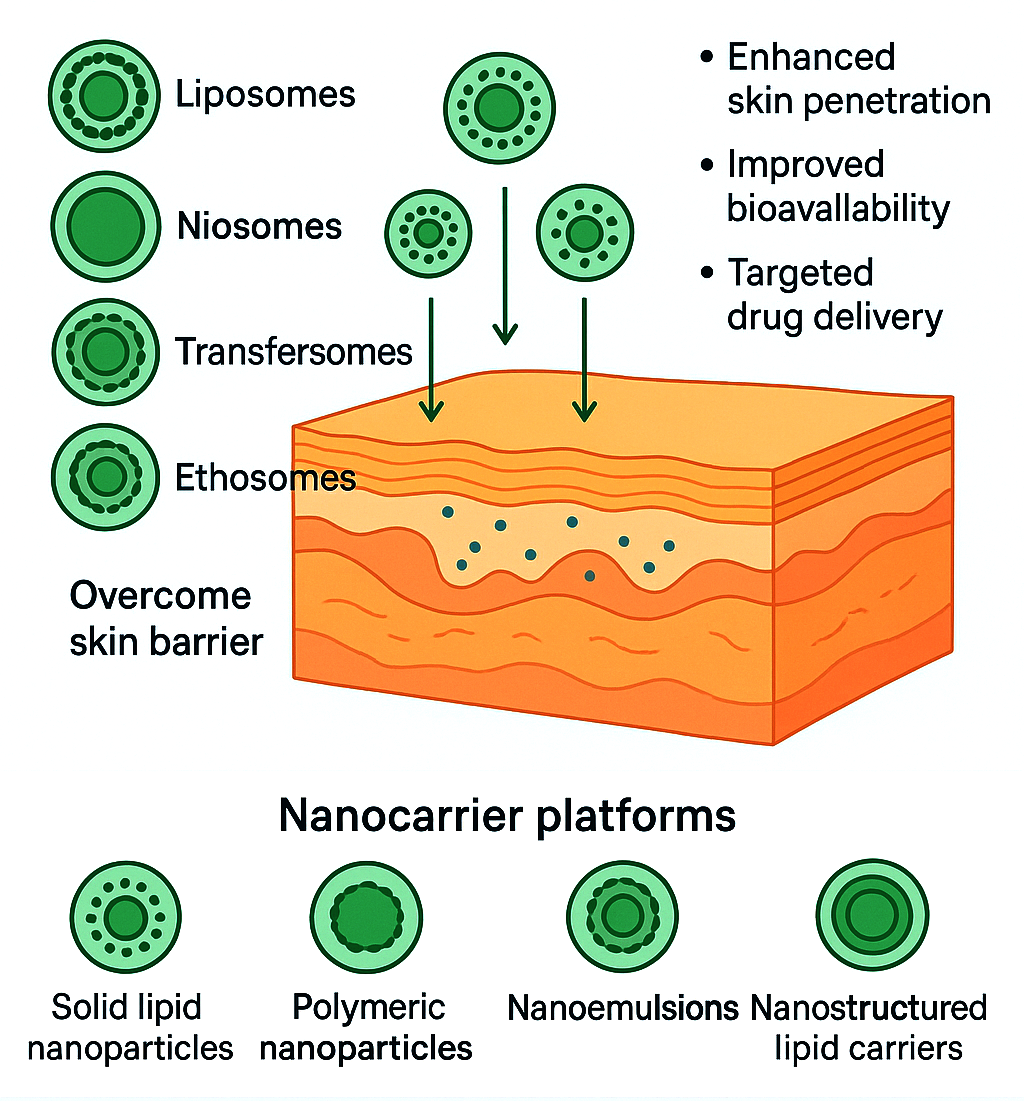
Transdermal drug delivery (TDD) offers a non-invasive, patient-friendly alternative to oral and injectable therapies by bypassing hepatic first-pass metabolism and providing controlled, sustained plasma levels. However, the stratum corneum represents a formidable barrier limiting drug permeation, restricting TDD to a narrow subset of therapeutically suitable agents and necessitating innovative delivery strategies. Nanomedicine has revolutionized transdermal drug delivery through nanocarrier platforms—including liposomes, niosomes, transfersomes, ethosomes, solid lipid nanoparticles (SLNs), nanostructured lipid carriers (NLCs), polymeric nanoparticles, and nanoemulsions—that enhance skin penetration, improve bioavailability, and enable targeted drug delivery. These nanocarriers exploit multiple complementary mechanisms, including lipid-mediated barrier modulation, transiently increased skin hydration, preferred routing through intercellular and appendageal pathways, and sustained, controlled release from the carrier matrix. Nanocarrier physicochemical properties—such as size, surface charge, elasticity, and composition—can be precisely engineered to optimize skin deposition, retention, and systemic absorption of diverse drug classes, ranging from small molecules to peptides, proteins, and nucleic acids. Clinical applications span pain management, hormone replacement therapy, cardiovascular disorders, and vaccine delivery, with demonstrated improvements in efficacy, reduced dosing frequency, and enhanced patient compliance compared to conventional formulations. Integration with advanced enhancement techniques—including microneedles, iontophoresis, sonophoresis, and stimuli-responsive triggers—further augments therapeutic outcomes and enables precision dosing. Challenges in large-scale manufacturing, formulation stability, batch-to-batch reproducibility, and regulatory harmonization persist, requiring continued innovation in formulation science and quality assurance. Future directions emphasize smart, stimuli-responsive nanocarriers, AI-driven optimization, personalized transdermal therapy, wearable device integration for real-time monitoring, and hybrid biomimetic systems. Collectively, transdermal nanomedicine represents a transformative paradigm, offering controlled, painless, and patient-centric drug delivery while expanding the therapeutic scope of previously unsuitable compounds—positioning nanomedicines as a cornerstone of next-generation pharmaceutical advancement and precision therapeutics.